Research
Over the last 20 years the BSRBR-RA study has contributed to a sizeable amount of drug safety research.
Here you can find the output and how you can access the work that is done at the register.
Contributors' list
We acknowledge all of the rheumatologists, nurses and allied health professionals who have ever contributed clinical data to the BSRBR-RA since it began in 2001. See the full contributors' list.
Accessible summaries
See lay summaries and infographics for some of our key publications.
Applying for a dataset
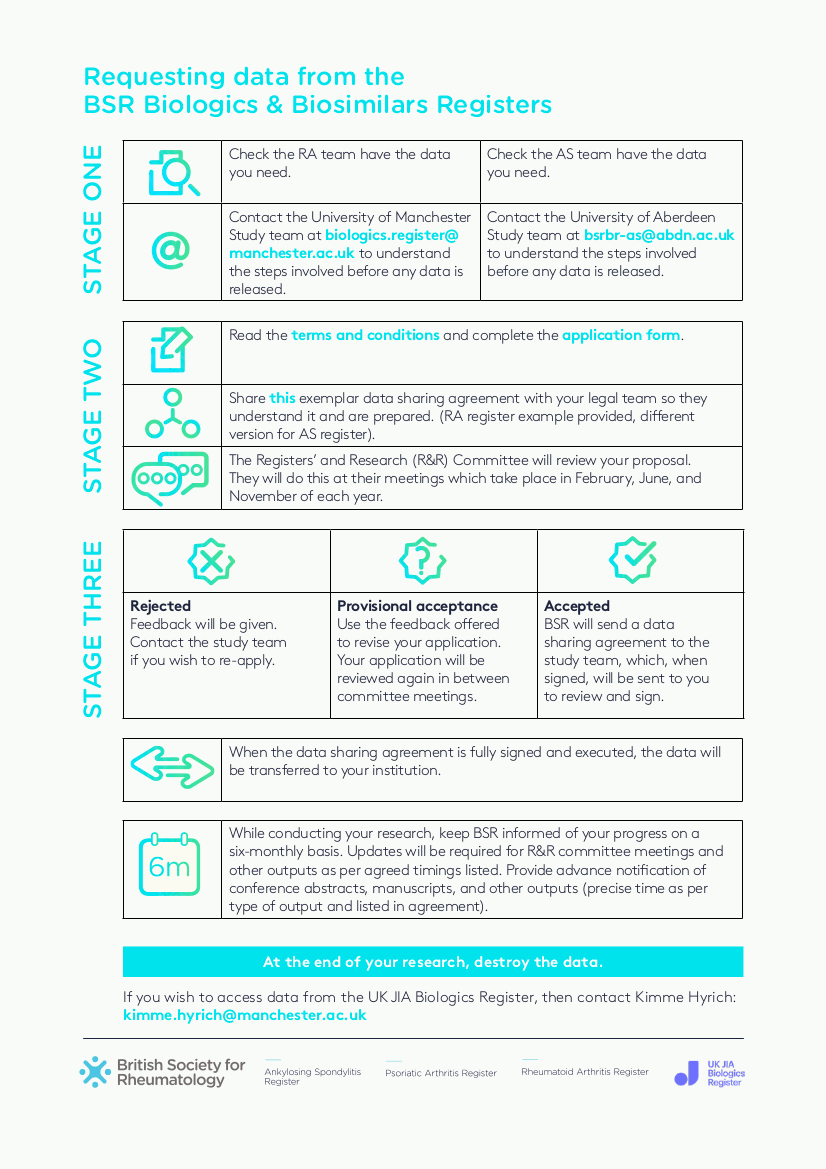
External parties are encouraged to access and analyse the rich BSRBR-RA data set. However, there are some contractual limitations on how the data can be used. All third party data requests are initially handled by The British Society for Rheumatology, following the process outlined in the infographic below:
[click on image to open as pdf]
Visit the BSR website for further information on requesting registers data.